|
What is the electronic configuration of Gold 79? What is the boiling Point of Gold in Kelvin?īoiling Point of Gold in Kelvin is 3129 K. Melting Point of Gold in Kelvin is 1337.33 K. What is the melting Point of Gold in Kelvin? What is the boiling Point of Gold?īoiling Point of Gold is 3129 K. Gold has 79 electrons out of which 5 valence electrons are present in the 5d10 6s1 outer orbitals of atom. How many valence electrons does a Gold atom have? Gold was first isolated by Varna Necropolis in ca. The element Gold was discovered by Bulgaria in year Before 6000 BCE. It is located in group 11 and period 6 in the modern periodic table. Gold is the 79 element on the periodic table. Gold is a chemical element with the symbol Au and atomic number 79. What is the position of Gold in the Periodic Table? Gold is a chemical element with symbol Au and atomic number 79. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Gold is 4f14 5d10 6s1. What is the abbreviated electronic configuration of Gold? What is the electronic configuration of Gold? Gold Thermal Properties - Enthalpies and thermodynamics Optical Properties of Gold Refractive IndexĪcoustic Properties of Gold Speed of Sound Gold Heat and Conduction Properties Thermal Conductivity Refer to table below for the Electrical properties ofGold Electrical Conductivity Hardness of Gold - Tests to Measure of Hardness of Element Mohs Hardness The value of m s doesn't really matter as long as there are no repeating values in the same orbital.Refer to below table for Gold Physical Properties Densityġ9.3 g/cm3(when liquid at m.p density is $17.31 g/cm3) The value of m l can be determined by some of the rules we will learn in the next chapter. Basically, the row tells you how many shells of electrons there will be, and the column tells you which subshells will occur (and which shells they occur in). Each row introduces a new shell (aka energy level). The bottom portion makes the f-block (Lanthanides and Actinides). 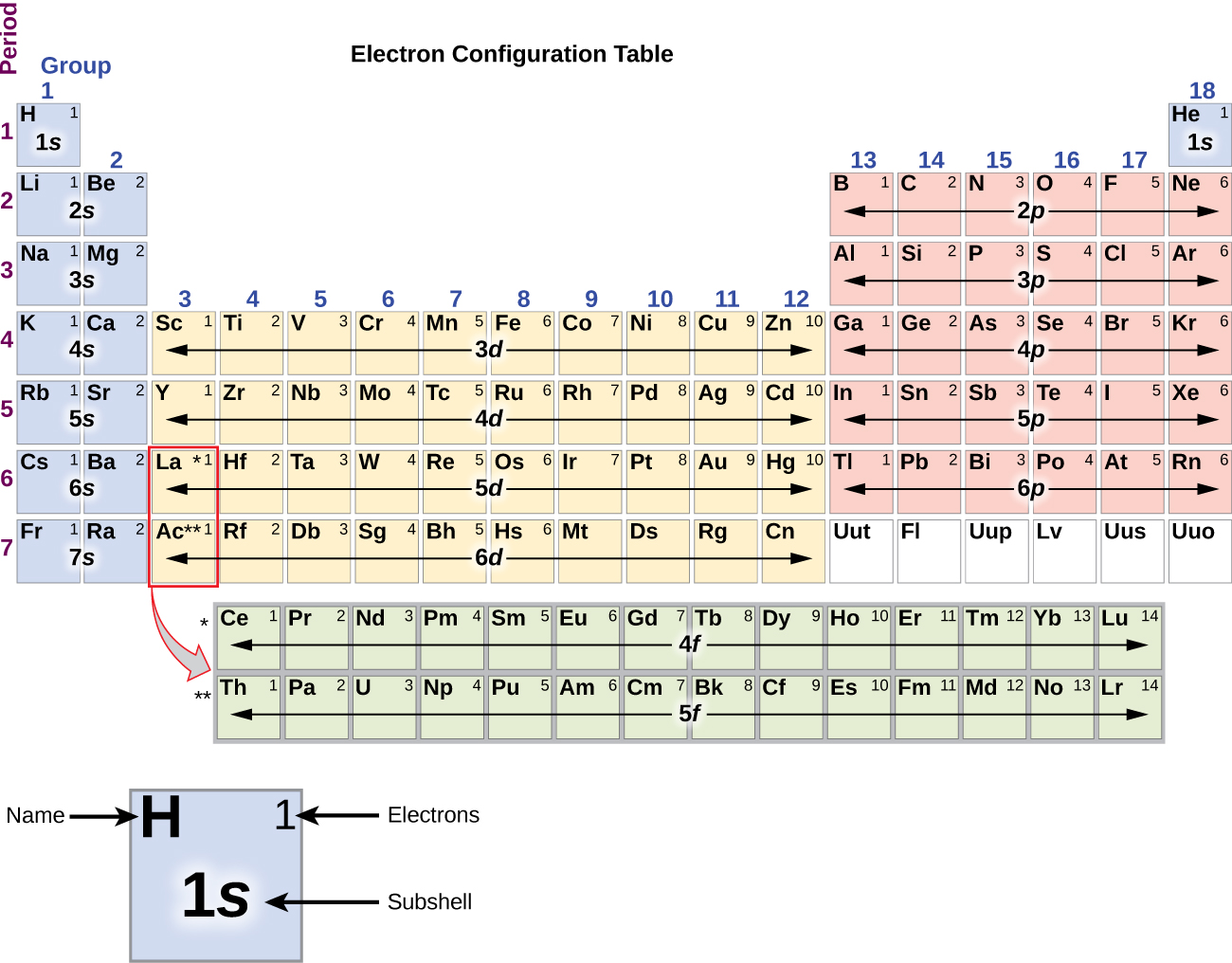
The large area in the middle (transition metals) makes the d-block. The six columns on the right make the p-block. The two columns on the left make the s-block. Different areas, or blocks, have different types of electrons. Remember that each list of numbers corresponds to ( n, l, m l, m s). Let's examine the quantum numbers of electrons from a magnesium atom, 12Mg. Every orbital can contain one "spin up" electron, and one "spin down" electron.  
The second arrow, or down arrow, is considered -½. If there is only one electron in an orbital (one arrow in one box), then it is always considered +½. +½ corresponds to the up arrow in an electron configuration box. Spin Quantum Number ( m s) ĭoes not determines the spin on the electron. These four numbers are used to describe the location of an electron in an atom.ġ, 2, 3, 4, … subshells are still degenerate regardless of shape differences.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
